What does the atomic mass represent? Read on to find out!
Atomic number and atomic mass
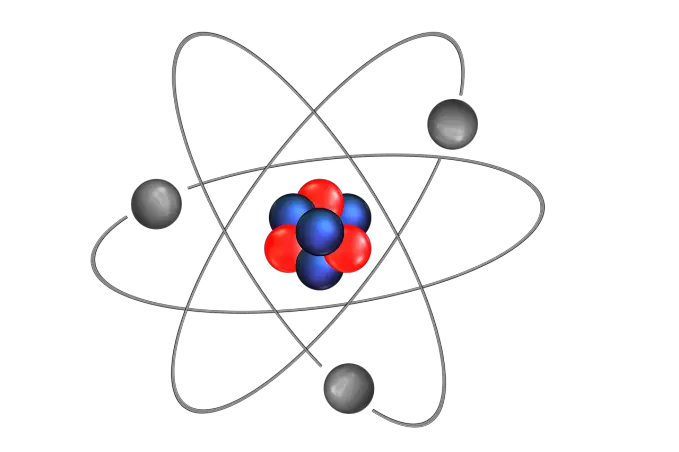
Although atoms are the smallest unit of matter, they are made up of even smaller particles. Each atom consists of a nucleus, made of protons and neutrons, and outer shells of electrons.
What is the atomic number of an element?
The atomic number of an element is the number of protons the atom has in its nucleus and, because each element has a different number of protons, this number determines the element’s identity.
The atomic number of an element is the number of protons the atom has in its nucleus.
The number of electrons is also the same as the atomic number, as the atom is neutral and therefore has equal numbers of positive protons and negative electrons.
More than one form of each element exists on Earth; these variants are called isotopes and differ from each other only in the number of neutrons each has in its nucleus.
When determining the mass of an atom, protons and neutrons are counted, and they have masses of approximately 1 atomic mass unit each. Electrons are negligible in atomic mass calculations as their mass is 1/2000th of an atomic mass unit.
What is the atomic mass of an element?
So, the atomic mass of an element is calculated by adding together the number of protons and neutrons an atom has. On the periodic table, the atomic mass of elements increases as the atomic number increases, with a few exceptions.
The atomic mass of an element is calculated by adding together the number of protons and neutrons an atom has.
The periodic table shows the atomic mass of each element, and it’s almost always a decimal. This is because the value shown for the atomic mass is actually a weighted average, determined using the proportional amounts of all the isotopes of that element on Earth.
Because there is usually one isotope that is much more common on Earth, rounding the atomic mass to the nearest whole number gives the atomic mass of that isotope, and is called the atomic mass number.
For example, on the periodic table, sodium (Na) has an atomic number of 11 and an atomic mass of 22.990. The decimal 22.990 indicates that there are multiple isotopes of sodium, each with a different number of neutrons.
However, the isotope with a mass of 23 is the most common by far, so the weighted average of all the sodium isotopes is very close to 23. Thus, the atomic mass is rounded to the nearest whole number, 23. Each atom of sodium therefore has 11 protons and 12 neutrons in its nucleus (11 p+ + 12 no = 23) and 11 electrons orbiting in its shells.