Why does water expand when it freezes? Read on to learn the answer.
Water is a polar molecule
Water is a polar molecule. Each atom on the periodic table has a characteristic called electronegativity, or the tendency of that atom to attract electrons.
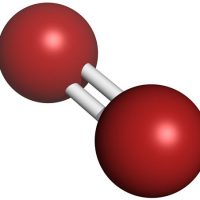
The atoms in water are covalently bonded, meaning the two hydrogens and one oxygen are sharing pairs of electrons (versus an ionic bond, where the electrons are completely donated from one atom to another).
However, they are not being shared equally. Because oxygen is more electronegative than hydrogen, electrons tend to spend more time near the oxygen atom than near the hydrogen atoms. This results in a polar covalent bond, where one pole of the molecule is slightly positive (the hydrogens), and one pole is slightly negative (the oxygen).
Because of its polarity, water forms bonds with itself called hydrogen bonds. The slightly positive regions of one molecule and the slightly negative regions of another are attracted to each other. Hydrogen bonds are very weak, but there are vast numbers of them, so they are strong overall. Now let’s consider how this helps answer the question of why water expands when it freezes.
Why does water expand when it freezes?
Water’s solid state, ice, is unique. It is the only substance that expands when it freezes. Normally, when a substance is cooled, it become denser. Its particles lose kinetic energy and can’t resist their natural attraction for each other. As they freeze, they pack tightly into crystals that are much denser than the same substance in liquid form.
Liquid water is a partially-ordered structure in which molecules are hydrogen-bonded to each other, but these bonds are constantly being broken and formed.
As it cools, water does become denser, reaching maximum density at about 4oC. At that point, however, the molecules begin orienting themselves in a very rigid lattice structure held together by hydrogen bonds. The crystals formed have a hexagonal shape that leaves more space between the water molecules than exists in liquid water. Other forms of ice that are more dense than water do occur, but they don’t form under natural conditions.
Thus, water expands when it freezes and ice floats on water. This causes large bodies of water such as lakes and rivers to freeze from the top down, and rarely all the way through, as the ice at the top insulates the water underneath. Living things can survive under the ice through winters and ice ages.