Nearly all the metabolic processes in the cell – the building up and breaking down of large molecules – are catalyzed by enzymes. Enzymes, like all catalysts, accelerate chemical reactions without being permanently altered. Because of this, they can be used over and over, making them very effective in small quantities. Enzymes catalyze several thousand types of biochemical reactions, allowing these processes to occur quickly enough to sustain life. Let’s now go into more detail as we answer the question, what are enzymes?
Enzymes are biological catalysts, usually made of protein. They speed up chemical reactions without being consumed or permanently changed, meaning they can be used repeatedly.
Enzymes explained
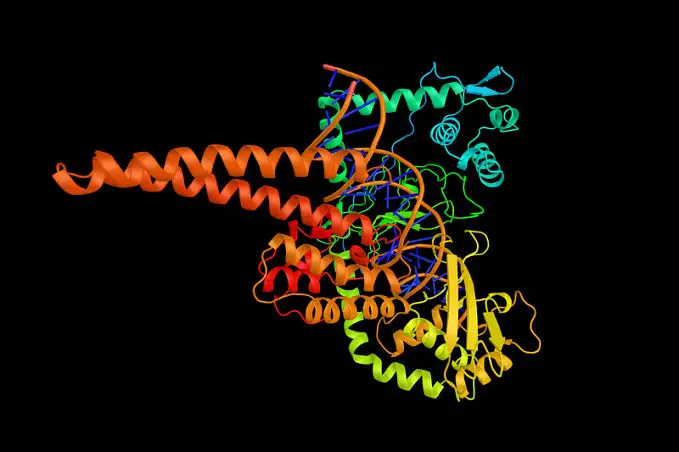
Nearly all enzymes are made of protein and have a very specific three-dimensional shape. This shape allows them to fit with the molecules they act on, their substrates, with perfect precision. Enzymes are substrate-specific, in that they only fit precisely with their own substrate. This means that each biochemical reaction has a dedicated enzyme.
All chemical reactions require some amount of energy input, called activation energy, to make the reaction happen. The energy is used to break the existing chemical bonds so that new ones can be formed.
In chemistry, heat is often used as a catalyst, as it gives the reactants enough energy to get the reaction started.
However, in cells, temperature is fixed, so reactions have a hard time getting started. Enzymes lower the amount of activation energy needed for the reaction to occur, but the end product is still the same. Like getting an expensive item at a reduced price, you pay less but still get the same product. This way, the activation energy can be achieved at cellular temperatures.
Most human enzymes function best at 37oC, body temperature. Lower temperatures slow reaction rates because there aren’t as many collisions between the substrates and the enzymes.
Higher temperatures destroy the shape of the enzyme with too many violent collisions. In terms of pH, most enzymes function optimally between 6 and 8, but some are better suited to extreme pHs, such as pepsin, which functions best in the very acidic environment of the stomach.
The site on the enzyme where the substrate bonds is called the active site. Changes in the cellular environment (e.g. pH) or interference from other molecules (e.g. inhibition) can alter the shape of the active site, essentially making the enzyme useless.
Inhibition is usually detrimental, for example carbon monoxide and cyanide, which both interfere with an enzyme and render it useless. But inhibition can also be used to control the metabolic pathway the enzyme is in.
For instance, large amounts of a product in a metabolic pathway aren’t practical if they can’t be used. But in some cases, molecules of the product itself can inhibit an enzyme in the metabolic pathway that produces it, essentially turning the production of itself off. As concentrations of the product drop, the enzymes turn back on, allowing more of the product to be made. This prevents the cell from wasting energy and resources.
Most cellular reactions would take place given enough time, but enzymes allow these reactions to occur quickly enough to support life. They also allow metabolic control and make cellular functions more efficient.