What is nuclear fission and what’s the difference between fusion and fission? Read on to learn the answers to these questions and more.
What is nuclear fusion?
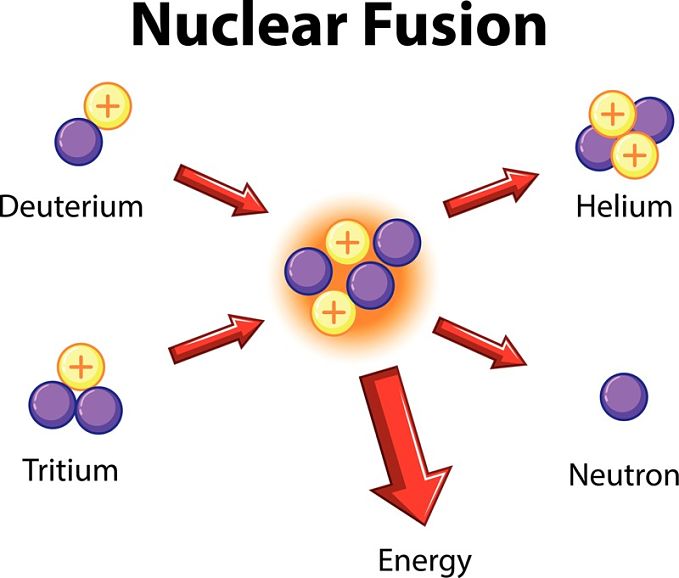
Nuclear fusion is a reaction where atomic nuclei are brought together in very close proximity so that they fuse together into a new nucleus.
Nuclear fusion explained
Nuclear fusion is a reaction where atomic nuclei are brought together in very close proximity so that they fuse together into a new nucleus. Because atomic nuclei are positively charged, they will repel each other, making fusion difficult.
However, there is another opposing force, the nuclear force, holding the protons and neutrons in the nucleus together. The key is overcoming the repulsive force so that the two nuclei can fuse together.
Nuclear fusion in the sun
Our Sun and other stars like it are powered by fusion. In the Sun, a series of reactions occur that fuse hydrogen atoms together into helium-4 atoms. The mass of helium-4 is about 0.7% lower than the four hydrogen nuclei that went into making it. According to Einstein’s famous equation, E=mc2, the missing mass is released as energy. In the Sun’s case, this energy is mainly heat and light.
The Sun is essentially an enormous nuclear reactor. With core temperatures at roughly 15 million K, and pressures at millions of times that on Earth, the Sun has ideal conditions for fusion to take place. However, these conditions are impossible to replicate on Earth in order to perform fusion reactions, which, until recently, has severely hindered the development of fusion as a source of energy.
Nuclear fusion on earth
The most common nuclear fusion reaction on Earth is the fusion of deuterium (hydrogen-2) and tritium (hydrogen-3). This reaction requires heating the reactants from a gas to a plasma at about 39 million degrees Celsius. The fusion reaction generates 17.6 million eV of energy, which is a very small amount, about 2.8 joules. However, a single gram of deuterium and tritium would produce 32 trillion times that amount.
Fusion reactions are only exergonic when using light elements. A large amount of energy is required to force nuclei to fuse together.
For heavier elements, the energy required to force the nuclei together is greater than the amount that is given off when the nuclei fuse.
But for light elements, the energy given off is greater, making them much more suitable for fusion reactions. Generally, if the nucleus created in the reaction is lighter than iron-56 or nickel-62, the reaction will give off energy.
Nuclear fusion reactions evoke images of mushrooms clouds and widespread devastation, and rightly so; a thermonuclear bomb, or hydrogen bomb, contains more than 500 times the explosive power compared to the atomic bombs that destroyed Nagasaki and Hiroshima in World War II.
However, it has great potential to be a future source of clean, abundant energy. Today’s nuclear reactors use atomic fission reactions, which generate more radioactive waste than fusion reactions and require materials that are far less available.
Deuterium, on the other hand, occurs naturally in ocean water, and about 4 litres of ocean water could produce the same amount of energy as over 1 000 litres of gasoline. As long as there is water on Earth, there could be a source of clean energy.