What is nuclear fission and what’s the difference between fission and fusion? Read on to learn the answers to these questions and more.
What is nuclear fission?
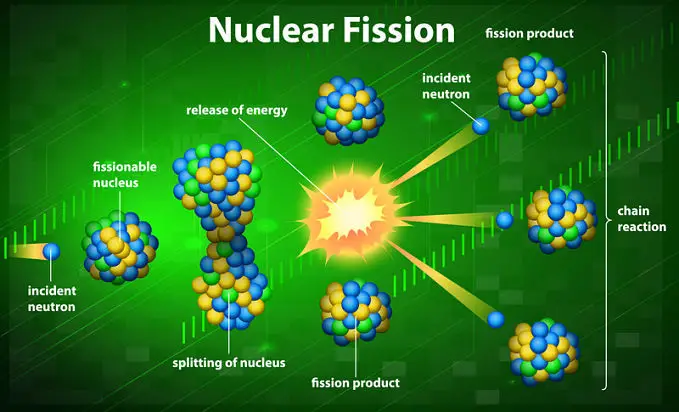
Nuclear fission is the splitting of a large atomic nuclei into smaller fragments, releasing energy.
Nuclear fission explained
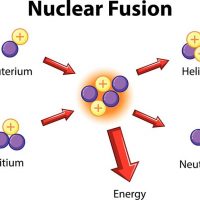
While nuclear fusion is the fusing together of two atomic nuclei into one large one, nuclear fission is the opposite: splitting a large atomic nuclei into smaller fragments, releasing energy.
The key is overcoming the nuclear force that holds the particles in the nucleus together, and forcing the nucleus to split.
This is a very exergonic reaction that releases a large amount of energy in the form of electromagnetic radiation as well as the kinetic energy of the fragments themselves.
Examples of nuclear fuels
The most common nuclear fuels are uranium-235 and plutonium-239, used in nuclear reactors.
The fission reaction is achieved when the nucleus of, for example, a uranium-235 atom absorbs a neutron, changing it into uranium-236. The addition of the neutron and its kinetic energy, along with the forces the bind the neutron cause the atom’s nucleus to split into lighter elements. These elements are roughly 95 and 135 amu (plus or minus 15 amu); other than the range of masses given, the products are unpredictable.
The fragments that are produced (there are usually two, called binary fission) are smaller in total size than the original atom. The difference in mass between the original atom and the two new fragments is released as energy.
As in fusion reactions, the energy released is equal to the mass difference times the speed of light squared, as given by Einstein’s famous equation, E=mc2.
Nuclear fuels that will undergo fission when hit with fast or slow-moving neutrons are called fissionable. Fissile nuclear fuels, a subset of fissionable fuels, are able to sustain a fission chain-reaction, where the initial fission reaction produces smaller atoms as well as neutrons that then trigger more fission reactions, which produce more neutrons that trigger more fission reactions, and so on and so forth.
This can produce energy at a controlled rate for nuclear power, and at an uncontrolled rate in nuclear weapons.
In a nuclear reactor, control rods are placed between fuel rods to absorb some of the neutrons produced by the fission reactions. These rods prevent the fission reactions from getting out of control.
Fission bombs (also known as atomic bombs or atom bombs), however, are designed to release as much energy as possible in as short a time frame as possible.
Hiroshima and Nagasaki
The most well-known example of an uncontrolled fission reaction is the pair of fission bombs dropped on Hiroshima and Nagasaki in August of 1945.
The bomb dropped on Hiroshima weighed 4 tons (of which 60 kilograms was nuclear fuel), was 3 metres long, and produced an explosion comparable to 15 000 tons of TNT – it wiped out 13 square kilometers of the city.
Almost 75 years later, a W88 warhead weighs less than 800 pounds, is 1.75 metres long, and can produce 30 – 40 times the explosive power as the Hiroshima bomb. A single missile can carry up to 12 of these bombs at once.
It’s easy to see how the power of the atom can be destructive. However, the amount of energy available in nuclear fuels is at least 10 million times greater than the energy in a similar mass of chemical fuel like gasoline, making controlled fission reactions a powerful source of energy for the foreseeable future.